This site provides INTERCEPT product information for International audiences Select your region
This site provides INTERCEPT product information for International audiences Select your region
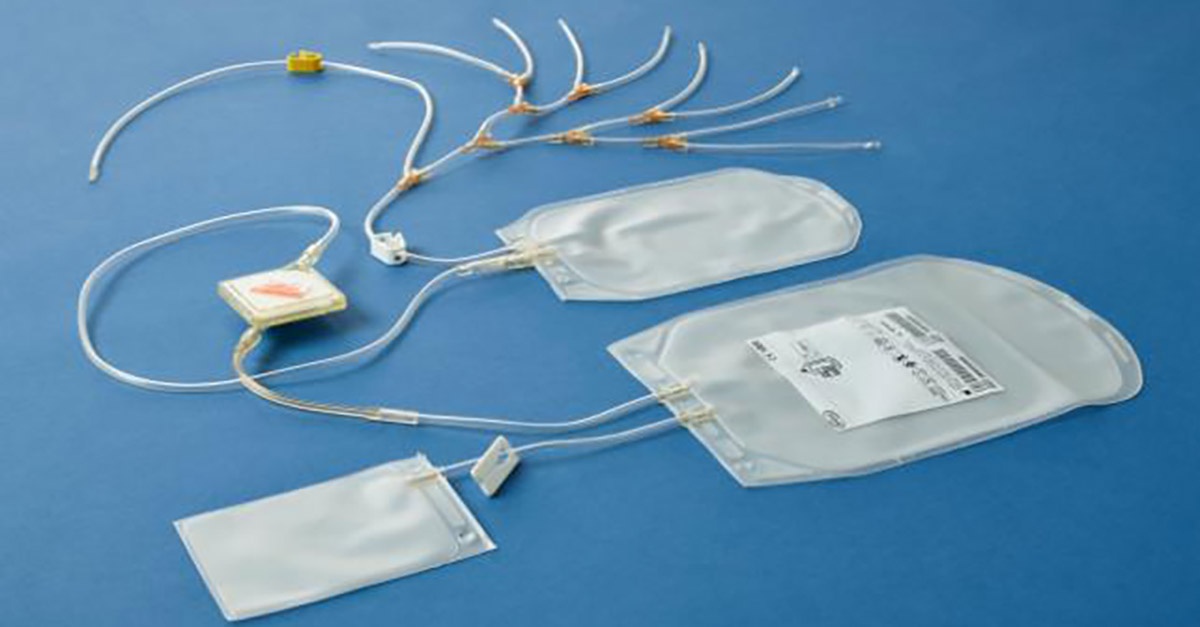
For blood centres seeking a cost-effective way to implement INTERCEPTTM pathogen inactivation, double dose buffy coat platelet production may offer a solution.
A platelet concentrate with sufficient platelet count to result in 2 therapeutic doses after pathogen inactivation with the INTERCEPT Blood System.
The double dose buffy coat platelet production process can contribute significantly to the affordability of the INTERCEPT Blood System for platelets, offering direct, as well as indirect financial and economic advantages.
There are several ways in which the pooling of 7 to 8 optimised buffy coats may contribute to the affordability of the INTERCEPT Blood System for platelets. In addition to a potential lower number of buffy coats required to obtain 2 therapeutic doses, the required number of platelet pooling sets, platelet additive solution (PAS) and INTERCEPT processing sets can be reduced by 50%, and the number of sterile docksrequired can be reduced by up to 30%.
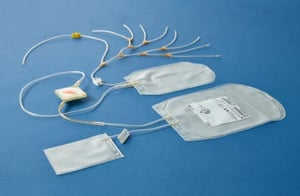 On top of these savings, the double dose buffy coat platelet production process can add to logistical and economic advantages: treating 2 therapeutic doses at once reduces the number of illuminations by 50%, equally saving on hands-on time. This, in turn, can translate into economic value.
On top of these savings, the double dose buffy coat platelet production process can add to logistical and economic advantages: treating 2 therapeutic doses at once reduces the number of illuminations by 50%, equally saving on hands-on time. This, in turn, can translate into economic value.
The I-Platelet Pooling Set is specially designed for double dose production of leuko-reduced platelets from whole-blood-derived buffy coat in additive solution and is validated to pool 7 to 8 buffy coats.
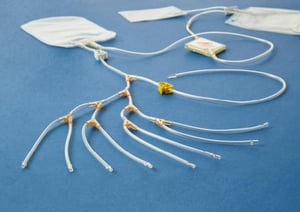
The total approach offered by Cerus relies on the skilled, well-trained and experienced Cerus Deployment team. They have developed a sound methodology to support blood centres in the implementation of the INTERCEPT Blood System for platelets in a cost-effective way.
The team members have extensive experience with successful implementations in blood centres of all sizes, transitioning from semi-automated as well as manual production methods. Based on this experience, they thoroughly assess the feasibility of the double dose buffy coat platelet approach in every blood centre, and assist with the implementation.
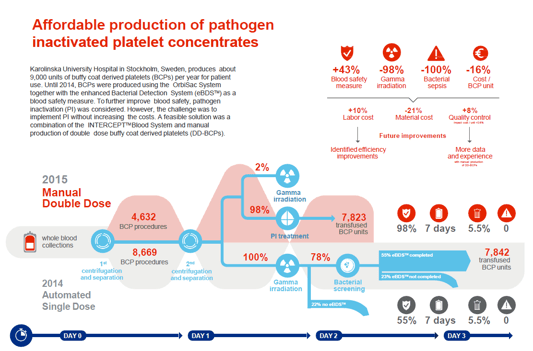
The Karolinska University Hospital in Stockholm (Sweden) is an excellent example of a blood centre that managed to implement pathogen inactivation in a cost-neutral, and long term cost-savings by switching from semi-automated single dose platelet production to manual double dose buffy coat platelet production.
eBDS is a trademark of Haemonetics.
Want to stay updated on the latest pathogen inactivation news?
You can receive our highlights by subscribing to our newsletter.
Do you have questions about the INTERCEPT™ Blood System, blood safety or blood transfusions? The INTERCEPT team has plenty of knowledge and expertise.
The information on this site is not country-specific, and may contain information that is outside the approved indications for the country in which you are located.
Use of INTERCEPT Plasma or Platelets is contraindicated in patients with a history or allergic response to amotosalen or psoralens. Consult instructions for use for indications, contraindications, warnings, and precautions.
Cerus, INTERCEPT and the Cerus logo are trademarks of Cerus Corporation.
© 2023 Cerus Corporation. All Rights Reserved. MKT-EN 00157 v43.0